A paywalled article in the British Medical Journal begins,
…the US National Institutes of Health infectious diseases chief, Anthony Fauci, appeared on YouTube to reassure Americans about the safety of the … vaccine. “The track record for serious adverse events is very good. It’s very, very, very rare that you ever see anything that’s associated with the vaccine that’s a serious event,” he said.
This was written in 2018; the YouTube appearance (below) was in October 2009, and the vaccine was the 2009 H1N1 Swine flu vaccine. Fauci addresses the risk-reward of the H1N1 vaccine starting just shy of the clip’s seven-minute mark. Much of his reassurances about the Swine flu vaccine will seem strikingly familiar to those who have been following the COVID-19 saga.
The BMJ article continues:
[B]y October 2009 the new vaccines were being rolled out … in the UK, with prominent organisations, including the Department of Health, British Medical Association, and Royal Colleges of General Practitioners, working hard to convince a reluctant NHS workforce to get vaccinated.
“We fully support the swine flu vaccination programme … The vaccine has been thoroughly tested,” they declared in a joint statement.
Except, it hadn’t. Anticipating a severe influenza pandemic, governments…had made various…arrangements to shorten the time between recognition of a pandemic virus and the production…and administration of that vaccine… [An arrangement], adopted by countries such as Canada, the US, UK, France, and Germany, was to provide vaccine manufacturers indemnity from liability for wrongdoing…
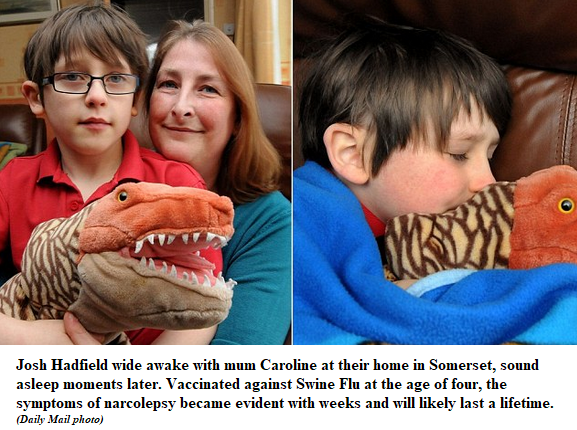
By 2018, the incidence of narcolepsy in young people throughout Europe as a result of vaccination, primarily with Pandemrix, was sufficiently well established to give rise to lawsuits in which the manufacturers’ confidential concerns about the vaccine were brought to light.
A 2012 article at orthomolecular.org, with the relevant citations in Swedish and Finnish, outlines the sequence of events (my emphasis).
On 25 September 2009, the European Medicines Agency (EMEA) approved Pandemrix … In Sweden, Finland, Norway and Iceland, the authorities explicitly set the goal of vaccinating the entire population…
Mass vaccination started in Finland and Sweden in October 2009. … [T]he authorities initiated an enormous public relations campaign… Solidarity became the slogan: “Be vaccinated to protect your fellow citizens.” … In Sweden, 60% of the population had been vaccinated, while in Finland 50% was covered.
In August 2010, Finland reported an increased occurrence of narcolepsy in children and youngsters vaccinated with Pandemrix. On 1 September 2010, Finland stopped all Pandemrix vaccinations.
On 1 September 2011, the Finnish National Institute for Health and Welfare (THL) … stated…, “The increased risk associated with vaccination amounted to six cases of narcolepsy per 100,000 persons vaccinated in the 4-19 age group during the eight months following vaccination. This was 12.7 times the risk of a person in the same age group who had not been vaccinated…
This statement was made almost exactly two years after the THL’s earlier statement…that it would be safe.
It will be pointed out, accurately, that on those figures from 2011 the risk of 4-19 year olds contracting narcolepsy from the vaccine was 0.006%. Keep that figure in mind.
In Sweden, at least 150 children are now [2012] suffering from narcolepsy caused by Pandemrix vaccine. In Finland, the number is approximately 100. … Narcolepsy is a disease with lifetime consequences, and the risk that Pandemrix may have caused other neurological illnesses has not yet been excluded.
By November of 2020, a Medical Xpress web article noted:
The Swedish Pharmaceutical Insurance has so far approved 440 of 702 narcolepsy claims linked to Pandemrix, paying out a total of 100 million kronor (9.8 million euros, $11.6 million) in compensation.
The current population of Sweden is 10 million; of Finland 5.5 million.
 In the UK, according to The Guardian in 2017, a High Court decision opened the way for “about 100 people in the UK with narcolepsy” allegedly caused by Pandemrix to claim compensation under the Vaccine Damage Payment Act. The Court rejected an appeal by the government to withhold payments.
In the UK, according to The Guardian in 2017, a High Court decision opened the way for “about 100 people in the UK with narcolepsy” allegedly caused by Pandemrix to claim compensation under the Vaccine Damage Payment Act. The Court rejected an appeal by the government to withhold payments.
In Ireland, however, the State admitted no liability in settling a 2019 case brought by a then 26-year-old woman. Another case was settled by mediation, without admission of liability, for €990,000. An Irish narcolepsy support group claims over 70 children are affected.
Narcolepsy sufferer Meissa Chebbi, ten at the time of her vaccination, is quoted in the Medical Xpress article. “I’m not going to take the (COVID) vaccine until after about five years when we know what the risks are.” This is the voice of bitter experience. The John Hopkins Coronavirus Resource Center puts it this way.
A typical vaccine development timeline takes 5 to 10 years, and sometimes longer, to assess whether the vaccine is safe and efficacious in clinical trials, complete the regulatory approval processes, and manufacture sufficient quantity of vaccine doses for widespread distribution.
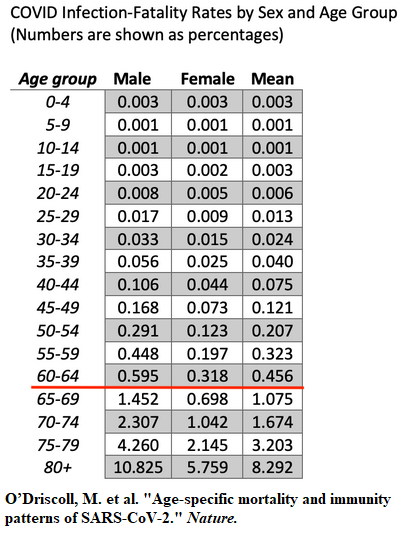
Just to get back to that extremely low risk of narcolepsy — 0.006% in 4-19 year olds. In April of 2021, the CDC’s own best estimate of the Infection Mortality Rate for the 0-19 years age group was three per 100,000 infected — 0.003%.
Peter West is a retired computer programmer. He contributed “The Willing Suspension of Disbelief” to the April 2019 issue and “The Burden of Proof and the Pell Case” in our March 2020 edition
 Sign In
Sign In 0 Items (
0 Items ( Search
Search










Thanks for posting. It’s nice to have evidence to confirm suspicions. I will share this widely, although many of my acquaintances may not want to hear this.
Good Youtube footage to unearth Peter. We need to be reminded of the health tragedies like this.
Here is Fauci sounding eerily familiar in 2009, when spruking the defective swine flu vaccination which was withdrawn, to his 2021 Covid 19 vaccination utterances.
This man seems a slow learner as he has repeatedly been shown to be wrong but never exhibits any doubt about his pronouncements. Who in their right mind would listen to this bloke given his track record?
On a related topic, I am of the view that Alan Jones is not coming back to Sky News (at least not with his previous format). The few times I did watch in the last 3 months or so, he was strong on the possible use and definite censorship of Ivermectin for C-19 therapy.
I caught Panahi just on closing last night where she was stating that Sky did not condone, nor would permit on air, any discussion of Ivermectin in the C-19 context.
End of story.
Overseas data is most useful for assessing the safety of the Covid vaccines because of the relatively large populations being treated. UK data is interesting in this regard. Dailysceptic.org publishes a regular Vaccine Safety Update based on UK Yellow Card data for the Covid vaccines. The figures are alarming.
Dr. Tess Lawrie, Director of the Evidence-based Medicine Consultancy in the UK, has recently written an open letter to the Director of the UK regulatory agency (MHRA) detailing the evidence for extensive adverse effects of the Covid vaccines. Her letter concludes:
“The MHRA now has more than enough evidence on the Yellow Card system to declare the
COVID-19 vaccines unsafe for use in humans. Preparation should be made to scale up
humanitarian efforts to assist those harmed by the COVID-19 vaccines and to anticipate and
ameliorate medium to longer term effects. As the mechanism for harms from the vaccines
appears to be similar to COVID-19 itself, this includes engaging with numerous international
doctors and scientists with expertise in successfully treating COVID-19.”
He is not a slow learner, just someone that has escaped imprisonment or worse for the time being.
Remember thalidomide?
Remember thalidomide?
“I will share this widely, although many of my acquaintances may not want to hear this.”
Earlier this year, when the vaccines were just becoming available for seniors, a medically qualified friend asked why I had decided not to agree to vaccination with the current CV19 vaccines.
I replied that vaccines require around 5-7 years, at least, to be adequately tested for longevity of safety. And added, “As you know.
” He nodded, without further comment. His wife abruptly rose from the table and joined another group. He followed.
Ceres. You ask why Fauci still has a job and why he is not in prison. There is a certain Tim Flannery with a similar record of zero correct pronouncements on climate yet is still the go to guy for all leftist media. If you are of the left as both these creeps are you do not have to be right but you should be be,believable by the unsceptical and gullible masses and you should be able to spruik whatever the elite wants you to spruik. It is preferable that you have no scruples, lack integrity and are not concerned about any damage you do to people or property.