
There is a considerable amount of misinformation, including bias by omission, in the mainstream media regarding COVID-19. As a pharmacologist involved in drug development, clinical trials and drug registration for 40 years – here are a few facts you need to know.
From where did Covid-19 come?
Coronaviruses can cause the common cold but there is little doubt that the coronavirus COVID-19 is a genetically engineered virus designed to be highly contagious. According to viral evolutionists, unique nucleic sequences have been inserted in this coronavirus, sequences which could not spontaneously arise by chance in nature. Since the beginning of this pandemic some leading infectious disease researchers and physicians and the World Health Organisation (WHO) have tried to claim COVID-19 came from the Wuhan wet market despite the absence of a single piece of evidence to support this proposition. Despite an intense search, no animal has been found to contain the virus.
There are many international patents which paved the way for the construction of COVID-19 based on work in association with the US National Institute of Health (NIH) and the US National Institute of Allergy and Infectious Disease (NIAID) using dangerous “gain-of-function” research.
Social media has de-platformed and censored anybody claiming the virus probably arose from the Wuhan Institute of Virology. Experts and the media are finally walking back the natural origin bat/wet market theory and admitting it is probable COVID-19 leaked from the Wuhan Institute of Virology in China. The implications of this are enormous as many people involved directly or indirectly in creating the virus (such as Drs. Anthony Fauci, Ralph Baric and Peter Daszak) now are involved in vaccine development and/or helping to set US government policy and shape public opinion while standing to gain financial benefit. Moore importantly, they count among some of the loudest critics of therapeutic measures other than vaccines to counter COVID-19.
These simple facts are the foundation for much of the misinformation and poor public health policy we are now experiencing. Until we clearly understand how this disaster happened we will be doomed to repeat the catastrophe.
The new vaccine technology
The new genetic vaccine technology used in the mRNA-based vaccine of Pfizer (and Moderna) and the DNA- based adenovirus AstraZeneca and J&J vaccines have never been previously approved for use as any new pharmaceutical. These vaccines could be called ‘genetic vaccines’ because they deliver genetic material for your cells to use as a template in manufacturing the identical spike protein which is found on the surface of COVID-19 virus. It is this manufactured spike protein, released into the blood from your cells following vaccination, which triggers your body to produce antibodies to protect you from a COVID-19 infection.
Have the COVID-19 vaccines been fully approved?
No.
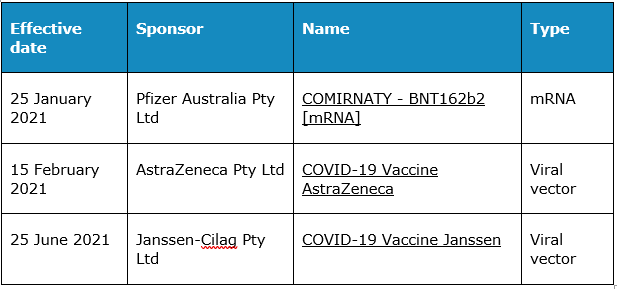
Due to the urgency of the pandemic, vaccines have been provisionally approved in the US and Australia pending the generation of further safety and efficacy information. The following COVID-19 vaccines (listed below) have been released in Australia under “provisional approval” which includes strict conditions such as the requirement to provide further long-term efficacy and safety information from ongoing clinical trials and post-marketing assessment from the reporting of adverse reactions.
Unresolved questions about the new vaccines
What is the duration of protection from infection following vaccination?
What is the chance of transmitting infection if one is fully vaccinated?
What is the risk/benefit of vaccinating individuals who have had COVID-19 and have developed natural immunity on their own?
Which vaccine is best for older people or young people?
Should all children, babies or pregnant mothers be vaccinated?
Are there circumstances where specific vaccines should not be used (contraindications)?
How effective are the new vaccines against emerging strains of Covid-19 such as the Delta variant?
How effective are the new vaccines?
The new mRNA vaccines were provisionally approved on the basis of a single randomised, controlled clinical trial involving about 40,000 volunteers. Regulatory bodies, such as the US Food and Drug Administration in the US and the Therapeutic Goods Administration in Australia, usually require many clinical trials for approval. However, this was an exceptional set of circumstances. Gaining approval after only a single clinical trial means that it becomes extremely important to gain additional post-market safety and efficacy data.
Now that millions of doses have been used, experience to date strongly suggests the genetic vaccines can prevent, to a large degree, more serious COVID-19 symptoms, hospitalisation and death, especially in those individuals with co-morbidities (age, diabetes, compromised lung and cardiovascular function).
It has been widely reported that the mRNA vaccines were about 95 per cent effective, but the question is: “effective at doing what”? In fact, the trials of the genetic vaccines did not find any difference in the death rates for vaccinated and unvaccinated groups (there were too few COVID-19 cases) and, furthermore, the primary criteria for being “effective” was a positive COVID-19 test and a symptom(s), including even a mild ones like a cough or fever. While this clinical trial result is one metric to indicate the level of immune efficacy, the selected criteria did not permit any reliable estimate of the level of protection against progressing to serious clinical conditions, being admitted to hospital or dying from COVID-19. This was because the numbers in these categories were too small to be statistically meaningful.
What does the daily reporting of “cases” tell us?
COVID-19 tests use long-established Polymerase Chain Reaction (PCR) technology. This test is so sensitive it can be set to detect even a single molecule of the target substance.
PCR tests measure a short nucleic acid sequence which is present in COVID-19 whether or not this is in the form of an intact COVID-19 virus or a fragment of a dead virus from a previously infected person. The test cannot tell if there is the presence of a live virus, nor can it determine the viral load (the amount of virus) in a positive test. Then why is the number of positive PCR tests the key metric driving public health policy in this pandemic, which is causing such massive economic and social damage? It makes no sense.
Given more than 99 per cent of people who are infected by COVID-19 experience either no symptoms or mild symptoms, shouldn’t our health policy be driven by the number of seriously ill subjects, patients admitted to hospital, or the number of deaths due to COVID-19 (not “with” COVID-19)?
Let’s keep a sense of perspective. There are normally more deaths expected due to ordinary pneumonia/influenza each year than have died with COVID-19 since the pandemic began.
How safe are the new mRNA and DNA vaccines?
Safety is a relative term in pharmacology. Anti-cancer drugs are highly toxic but they can also save your life. There is always, with any drug, an assessment of risk vs benefit. The benefit side of the equation is becoming clearer with widespread vaccine usage, but the risk side of the equation is far from clear because there has been no detailed, publicly available overall analysis of the incidence of adverse drug reactions (ADRs) that are likely or, probably, related to the vaccines.
The evaluation of ADRs includes a detailed analysis of the subject’s medical history, the circumstances surrounding the ADR, the temporal relationship to the drug and co-administered drugs, and consideration of the mechanism of action of the suspect drug. There are various guidelines and definitions used to estimate whether or not there is a causal relationship. It takes experts considerable time to assess with any precision which serious adverse effects observed are actually due to a drug’s administration.
In the past, safety concerns have led to the withdrawal of certain vaccines following widespread usage, and regulatory agencies routinely monitor the safety of vaccines via various ADR systems. There is a specific ADR system used by the US Centres for Disease Control (CDC), the Vaccine Adverse Event Reporting System (VAERS).
As a general rule, as few as ten per cent of ADRs are reported. This observation, coupled with the government’s perceived need to overcome vaccine hesitancy in the population, work to minimise ADR reporting and underestimate the true incidence of any particular ADR. This frustrates an accurate assessment of risk.
For mRNA vaccines, it has been reported that nearly one-third of those deaths reported in the ADR system occurred within two days of vaccination. These reports and thousands of hospitalisations following vaccination need full investigation to determine if there is any causal association. To date the public has not been appraised of any comprehensive, reliable and detailed analysis of the type and incidence of adverse reactions likely to be associated with genetic vaccines given the worldwide experience.
Claims that the vaccines are “completely safe” should be considered premature until further safety data becomes available. Until that time, the risk-benefit proposition in healthy young people, those previously infected with COVID-19, infants, children and the pregnant remain unresolved.
Every vaccine is different. The use of more traditional vaccines, like Novavax in the near future, may present a relatively more attractive risk-benefit position, but only time will tell.
Masks
There is much misinformation about face masks.
First, there is no credible evidence in the literature that cloth models or the ubiqitous blue-paper surgical masks prevent to any significant degree the spread of airborne viruses. N95 masks can be effective, but they need to be fitted so there are no air gaps. If you do use a paper surgical mask of the common blue type, do not reuse it, do not place it on hard surfaces, do not put it on and off repeatedly — it is designed for aseptic single use. Surgeons use masks to prevent the infection of open surgical sites. They are not used to prevent the surgeon from getting an infection. There has been a dramatic increase in bacterial pneumonia worldwide associated with the use of masks mandated by governments. As to wearing masks outdoors, most authorities consider outdoor transmission of COVID-19 as improbable.
Recommending that very young children wear paper or cloth masks, given the incredibly low relative risk, has been the subject of experts’ criticism due to the deleterious effects on their physical and mental health. Apart from anything else, small children tend not to wear masks correctly and are most unlikely to follow aseptic procedures.
Delta variant
Various medical bureaucrats and politicians have exaggerated the threat posed by the Delta variant now sweeping the world. Multiple sources report the Delta variant to be more infectious but less deadly. Claims that the Delta variant is a “beast” that will kill thousands are not supported by current knowledge and contribute without justification to the creation of unnecessary fear.
Challenges of a vaccine-centric strategy
There are several problems in relying almost exclusively on vaccines to deal with the COVID-19 pandemic. Theoretically, putting all one’s eggs in a single basket seems to be a risky strategy when there is so much yet to be learned about the safety and efficacy of these vaccines, and because so much depends on a successful outcome.
At this point in time we should be spending more effort in considering preventative and/or drug treatment strategies for ambulatory patients through to seriously ill COVID-19 subjects. The advice to those who have been determined as COVID-19-positive is to isolate. No treatment is actively recommended – just wait until you feel better or become so ill you need to be hospitalised.
Alternative therapeutic strategies – hydroxychloroquine and ivermectin
Given the sweeping global impact of COVID-19, it would seem prudent to hedge our bets and pursue alternative therapeutic strategies in parallel with mass vaccination. Some conventional drugs, including corticosteroids to counter the inflammatory effects of COVID-19, as well as newer approaches, such as antibody infusions and remdesivir (an expensive and provisionally approved antiviral drug) have been shown to have limited efficacy but they fall short of a complete solution.
Certain other candidate therapies, including hydroxychloroquine and ivermectin, have been studied in many clinical trials and used in national programs in India, Mexico and South America with reported success. But there exists broad censorship across social media, mainstream media and the professional literature which is preventing the sharing of clinical trial results and the academic exchange of views. Up until recently this censorship has been further entangled in the stifling of debate regarding the origin of the COVID-19 virus.
On March 24, 2020, our TGA restricted the prescribing of hydroxychloroquine, citing concerns about media reports of increased “off-label use” (prescribing for other than officially approved uses) which might confront patients for whom it is essential in the treatment of malaria and certain chronic autoimmune diseases (including rheumatoid arthritis and lupus erythematosus). This ruling denied Australian doctors the option to prescribe hydroxychloroquine for their patients in relation to COVID-19. A surprising and unprecedented move, the TGA edict came with a warning about the “well-known serious risks to patients, including cardiac toxicity (potentially leading to sudden heart attacks)….” Even though this old drug has been widely used for decades, continues to be used by millions of people around the world, and is on the World Health Organisation (WHO) List of Essential Drugs there has been no prior enforcement of any such restrictive prescribing.
The TGA’s ban also raised concerns about interference in the sacred doctor-patient relationship. It is not uncommon for medications to be used “off label”, with doctors employing their discretion in prescribing a drug which they deem most suitable. In Queensland, early in the pandemic, prescribing hydroxychloroquine for COVID-19 was criminalised , with heavy fines and up to six months’ imprisonment available to be imposed. This, too, is a step without precedent.
The TGA has issued a detailed statement regarding the basis for its action (August 26, 2020) and the clinical evidence upon which the policy decision was based, including a review by the National COVID-19 Clinical Evidence Taskforce and reference to the previous and disappointing interim results of international trials of hydroxychoroquine. Also cited were regulatory actions in relation to hydroxychloroquine by the US FDA and the UK Medicines and Healthcare Products Regulatory Agency (MHRA). However, since that time considerable additional clinical evidence has been generated in regard to hydroxychloroquine. (Note: the sister drug to hydroxychloroquine, chloroquine, is not marketed in Australia.)
More recently, another drug, ivermectin, used to treat parasitic infections such as scabies, has been studied in clinical trials against COVID-19 and received attention as a possible useful therapeutic. Some published clinical trial data suggests ivermectin might be preferred to hydroxychloroquine because it may have an additional role in the late treatment of COVID-19.
Ivermectin is a compound with known antiviral and anti-inflammatory properties which appears to act by interfering with entrance into cells and their replication of mRNA viruses such as COVID-19. It was originally derived from unique natural compounds found in a bacterial culture near a Japanese golf course in 1975. Since its approval more than 40 years ago it has had a dramatic impact on human health worldwide, first being used in treating river blindness. Its effective, broad spectrum, safe, well tolerated and easily administered characteristics were employed to treat a variety of parasitic worm infections which blighted the lives of billions of poor and disadvantaged peoples in the tropics. Its discovers, Prof. Satoshi Omura and Irish biologist William Campbell, were awarded the Nobel Prize in Medicine in 2015, reflecting the magnitude of their achievement.
A comprehensive meta-analysis of 18 randomised and controlled clinical trials regarding the efficacy of ivermectin in the prophylaxis and treatment of COVID-19 has been published in a prestigious medical journal, The American Journal of Therapeutics 28 (e299-e318, 2021), which announced statistically significant reductions in mortality, progression to serious disease and time to clinical recovery as well as preventing COVID-19 infections (prophylaxis). Another comprehensive meta-analysis of 24 randomized controlled trials involving 3406 participants reached similar conclusions (American Journal of Therapeutics 28, e434-e460). Randomised (where subjects are randomly allocated to a treatment group) and controlled (a comparison treatment group) clinical trials are considered the most reliable and least-biased clinical trials, while meta-analyses can be a powerful tool with which to analyse clinical trial data as it does not depend on the individual results of any one study but, rather, the sweep of results from a much larger group of patients under a defined set of rules, even though under somewhat diverse, but well defined, conditions.
A discussion of the safety and efficacy assessments of hydroxychloroquine and ivermectin, important as it is, is beyond the scope of this essay. However, it should suffice to say that there exists considerable international expert support, particularly for ivermectin, for the prophylaxis and treatment of COVID-19 based on the results of published randomised, controlled clinical trials.
Widespread social and professional media censorship of the debate and data concerning therapeutic management using hydroxychloroquine and ivermectin has not assisted an informed position regarding these drugs. According to the TGA’s latest advice (July 5, 2021):
There is currently insufficient evidence to support the safe and effective use of ivermectin, doxycycline and zinc (either separately, or in combination) for the prevention or treatment of COVID-19. More robust, well-designed clinical trials are needed before they could be considered an appropriate treatment option. The National COVID-19 Clinical Evidence Taskforce, consisting of a large group of clinical experts, is continuously updating treatment recommendations based on the best available evidence. They have not made any recommendations for the use of ivermectin, doxycycline or zinc outside of properly conducted clinical trials with appropriate ethical approval.
The use of ivermectin for COVID-19 in Australia is prevented by effectively blocking its supply and dispensing. The public trusts expert bureaucrat medical and public health opinion (which involves the TGA and expert committees), so opposing voices challenging current official advice on COVID-19 treatment struggle to have an impact in our current environment of information suppression, a gag never before seen.
I refer specifically to the Australian voices of Prof. Robert Clancy (Emeritus Professor of Immunology, Univ. of Newcastle) and Nobel Prize winner Prof. Thomas Borody, also echoed by Craig Kelly MP (Member for Hughes, NSW), who have argued to broaden the treatment strategy for COVID-19, expanding it from an approach dependent almost exclusively on mass vaccination to one involving the treatment, especially the early treatment, of ambulatory symptomatic subjects with repurposed drugs such as ivermectin.
On the one hand, it seems the TGA can lower the data bar for the interim approval of the new vaccines but it appears not to be so compromising in relation to the level of data required for a drug like ivermectin. This seems to be somewhat out of character because there have been circumstances in which the TGA has demonstrated considerable flexibility where it was important to do so.
I specifically refer to a case with which I was involved – botulinum toxin (now marketed as Botox). Botulinum toxin is one of the most deadly neurotoxins known to man but was approved without a single randomised controlled clinical trial for the rare condition termed blepharospasm (uncontrolled eye blinking). That was a good, but surprising, decision by the TGA. By comparison, there is an enormous amount of randomised, controlled clinical safety and efficacy data to support ivermectin.
Conclusions
As the vaccine rollout continues in the fight against COVID-19 we are gathering important new information with which to assess and define the safety and efficacy of the new vaccines and the risk-versus-benefit in various subgroups of the population. But until we can accurately define many unresolved questions regarding vaccine safety and efficacy, it would seem unwise to pursue a one-size-fits-all strategy. It would appear prudent to cover our bets by exploring the usefulness of drugs such as ivermectin which have shown encouraging results, are cheap, have been used safety for decades and are readily available. We should not waste any more time.
Disclaimer: The information contained above does not represent health advice
 Sign In
Sign In 0 Items (
0 Items ( Search
Search









For me, the scariest sentence in this essay is “According to viral evolutionists, unique nucleic sequences have been inserted in this coronavirus, sequences which could not spontaneously arise by chance in nature.” If this virus didn’t occur naturally, then it was let out into the world either deliberately or by accident. Given the strict conditions under which these labs operate, then deliberately gets the nod IMO. One wonders too if the strenuous efforts on the part of bureaucracies all over the world to cancel anyone who suggests that ivermectin or hydroxychloroquine might have a positive effect in the treatment of this disease, quite apart from barracking for big Pharma, might have something to do with being anti the previous President of the USA, who actively supported these products. Maybe after the current President’s most recent performance at a town hall meeting (https://www.youtube.com/watch?v=rSaTXfeKFvA) people might start realising that the previous one was light year’s ahead of the current one, and maybe was worth listening to.
Ivermectin and hydroxychloroquine have been demonstrated unequivocally to be effective for prophylaxis and early treatment of Covid-19 in numerous studies. Their use is supported by highly experienced physicians in many countries. The American Association of Physicians and Surgeons (AAPS) and the Frontline Covid-19 Critical Care (FLCCC) Alliance have developed sophisticated multidrug protocols designed to address each element of this complex disease in its life-threatening, acute phases. These protocols have been shown to dramatically reduce hospitalisation and death from Covid-19.
Dr. Kory and Dr. Marik of the FLCCC Alliance are among the most experienced and highly-credentialed critical care physicians in the world. Why are their treatment protocols banned in Australia? It seems that the interests of sick and dying patients are being sacrificed at the behest of powerful vested interests: the drug companies and the politicians are determined to let nothing hinder their mass vaccination program. The ready availability of cheap, effective treatments using harmless repurposed drugs would render mass vaccination for Covid-19 superfluous. Given the mounting toll of adverse events and deaths from the experimental vaccines now in use, an objective observer might conclude that banning Ivermectin and Hydroxychloroquine for Covid-19 is both irrational and unethical.
The FDA has come out in the last few days and announced that the CDC PCR test (used here) for COVID-19 has failed its full review. Emergency Use Authorization has been REVOKED. A class 1 recall which is the most serious.
Appears there are questions about its ability to differentiate between SARS Covid 2 and influenza viruses.
This test is used to determine positive cases and is what draconian lockdowns in Australia are predicated on.
Very interesting also, given that the 40 cycles used here has been considered too high. Apparently many false positives so this latest development adds to the questions about the precision of the test.
As I read the announcement – Laboratories will have until the close of 2021 before the CDC officially withdrawals its Emergency Use Authorization of the Real-Time RT-PCR Diagnostic Panel.
withdraws!
Given the TGA’s recalcitrance regarding substantive evidence for positive outcomes using multi-drug therapies based on Ivermectin and Hydroxychloroquine, there seems to be only one damning conclusion about the TGA’s relationship with Big Pharma.
Would you describe someone who chose not to purchase a new car equipped with an unconventional engine, that had no track record for reliability or safety and which was sold “as is,” without a warranty of any kind and made by a company that had somehow wrangled legal immunity from being sued if the car turned out to be a dangerous lemon, and produced by a company with a history of fraud as “hesitant” – or prudent?
Rod, sounds like someone who would buy a Tesla.
rod.stuart
How would you describe politicians who recommend, bordering on order, people be injected with a substance produced by a company with this rap sheet? False claims, kickbacks and bribery….
https://violationtracker.goodjobsfirst.org/parent/astrazeneca
Has it occurred to anyone else that the following five bits of information, (whether false or not) might be connected?
a) Since the late 1970’s the UN has been trumpeting Malthusian ideology first as Agenda 21, the Agenda 2030, in which the population is to be restricted to far fewer people than at present, and crammed into a few high density metropolises. In the last decade, the WEF as well as other dangerous misfits preach “Build Back Better” and a world population of about 2 billion people.
b) The Covid 19 shambles from the very beginning has clear ties to the Davos crowd and the WEF, as well as other shady operations such as the Bilderbergs and the Trilateral Commission and Council on Foreign Relations. It is becoming obvious to many that the objective of the scam is to inject as many people as possible with the shit shot.
c) A growing number of very knowledgeable people are questioning the long-term effect of this unknown experimental injection, and are concerned that it may result in multiple deaths of the next two or three years.
d) The Deagel organisation, closely associated with the CIA, a few months ago forecast the population of a variety of countries the world over in the year 2025. While the population of China, Russia, and many other countries remains the same of declines slightly according to this forecast, the population of the USA is reduced by two thirds, Australia, New Zealand, and Canada by half with similar declines in other Western countries. A flurry of conversations about this forecast on social media a few weeks ago resulted the forecast being taken down from the Internet.
e) The WEF, Johns Hopkins, and the Gates Foundation organised Event 201 which simulated a pandemic and preceded the release of the Wuflu. On July 9 this same cabal organised Cyber Polygon effectively simulating widespread hacking up to shutting down the Internet. Was this a precursor to the next planned calamity?
I’m too old to lose too much sleep over this, but it is food for thought.
https://vimeo.com/578965489/41cdfa68b6
May i suggest starting about 5 minutes 30 seconds in.. about the 6 minute 40 second mark is fascinating.Mc Nulty says” all except one are vaccinated “
Losthope, a word to the wise: Mcanulty — whose name is commonly misspelled as ‘McNulty’ even by AAP — subsequently resumed the microphone and said he misspoke, explaining that he meant to say ‘only one’patient had been vaccinated.
https://www.youtube.com/watch?v=xmqcDfwgkJs
Go to the end of the clip above for the correction
Prediction: the CDC’s withdrawal decision on the use of the PCR test for C-19 (wherein the test may not differentiate between C-19 and the annual flu strain) will NOT be accurately reported by the Aus MSM, if at all.
Social meeja will likely censor commenting on this development as being against community standards.
Thanks for pointing that out losthope.
Safe and effectiv, eh?
SURE it is.
Ceres..
you might be interested to know that apparently a defined ‘disease causing’ (my words) concentration of viral fragments is 5000 fragment per micro litre.
It seems PCR machines are set (at that 40 reps I suppose) to detect 4 fragment. Four.
Got that from this interesting video
https://www.questioneverything.io/replay/
FYI
A few days ago, I posted a comment on The Australian on-line edition in reference to one of their articles.
FULL TEXT :
” The question remains…why is not the Ivermectin protocol developed by Professor Thomas Borody discussed ( or implemented )”.
That’s it.
It was not published; but REJECTED.
……as I discovered when viewing ” MY Profile”
Scary stuff.
Feiko Bouman
How to promulgate this article? There appear to be no ‘share’ links and no ‘print version’ or ‘print to pdf’.?
I just wrote to the Premier of SA – replied to his broadcast email to us all – asking him how he feels knowing that if we suffer from covid because we couldn’t get ivermectin because he’s forbidden doctors to use it and the govt is complicit in making it unavailable elsewhere – we’ll be cursing him as we suffer?
And if we should die of covid we’ll be cursing him with our dying breaths?
I feel that’s fair enough. Why don’t you all send the same question?
Even if you don’t believe in Ivermectin. Isn’t it the same truth, the same question, with regard to the government’s blanket ban and silence on all alternative treatments, all self help, all intercourse with the people?
Isn’t it really the same for all? We’re being treated like lab rats. Well, worse, lab rats are at least provided for until they die. We, being people, are expected to provide for ourselves while they experiment with us.
I have a problem with being instructed to get a vaccine if I want to be “free” yet cannot sue the doctor who recommended it, administered it nor the company that made it if I get sick as a result. It goes without saying that public service types and politicians will absolve themselves from all responsibility. Knowing that the socialists and “save the world types” are strongly promoting the jab should give pause to everyone since they also want fewer people on the planet. They could volunteer to go first but seem to prefer others to be sacrificed. And we know they are hypocrites. Would you trust Hazzard and Gladys with your life????????????????????
If you think MSM will contribute to the propagation of the truth…
They are either heavily invested in pharma or get a lot for advertising so are in their pockets. Even if some brave journalists wanted, they stand no chance.
MSM, which in some countries are now called “main sewage media” are definitely not on the side of the truth, rather on the “right side of ‘history’ ” – that should be on the left side…
Pablo07, I prefer ‘Zombie Media’.
A few thoughts
Mask wearing.
At the beginning of the month I took one of my children into RPA and because of circumstances was allowed to stay with her.
The mask protocols were tight, people were checked in and had little dots of colour, mine was NAIDOC week.
Not all masks worn were highly effective against viruses…
A week later two infected people came in.
The outbreak never got anywhere.
Whatever masks do, they are a very ordinary part of medical and surgical practice.
They don’t protect in all cases.
From my limited observation they confer a benefit.
Arguing that they should be better reminds me of the adage
‘We need not make perfection the enemy of the good.’
The TGA statement above is at least surprising.
I have no expertise in treatment of humans.
The model for SARS/Covid respiratory disease is the cat.
High doses of doxycycline are routinely used in cats at 5mg/kg twice daily by mouth. It kills deep infection of the lungs and also, at that dose rate, is an anti-inflammatory.
There are case studies of the use of doxy in high risk human patients in the literature with Covid and comorbidity.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7476338/
Note that the doxy dose rate was high.
This does not align with the above statement by the TGA viz
‘There is currently insufficient evidence to support the safe and effective use of ivermectin, doxycycline and zinc (either separately, or in combination)’
What evidence do they need that Doxycycline is of no use?
Not sure about ivermectin.
It works in vitro.
The major support comes from its mass use in places like India and Brazil.
However these countries have endemic levels of migrating parasite pathogens susceptible to ivermectins, due to closeness to the soil and poor sanitation.
Hence the ivermectin may be treating the co morbidity, not the Covid.
https://pubmed.ncbi.nlm.nih.gov/12785174/
Its quite possible to lump a whole lot of trials or cherry pick them and get the good outcome.
But they have to be controlled for the particular population.
Australia is not India or Brazil.
Veterinary trialling is fraught with error so is Human.
For years vets used ace inhibitors to treat congestive heart disease in dogs.
‘The science was in.’
But two further longitudinal trials show it has no benefit.
A few specialists use it in the final stages of heart failure.
Zinc
Zinc in too high a dose is a heavy metal poison.
In animals the body needs zinc to heal.
So animals with adequate zinc are healthier than those that are not.
The primary response by an animal to septicaemia of bacterial origin is to run a temperature, curl up and go to sleep and strip zinc from the circulation.
The latter deprives the bacteria the substrate to grow.
Adding zinc would deprive the animal of this defense.
In long term high risk cases such as my daughter’s , after 4 months of parenteral feeding, the trick was to make sure that zinc and magnesium levels were adequate prior to surgery, so she healed.
So a one size fits all and the blind administration of zinc may actually be bad, as it could promote heavy metal toxicity, prevent native defenses against septicaemia and secondary bacterial pneumonia while not necessarily fixing the covid.
Vaccination.
There is a lot said about how these vaccines are novel and not adequately trialed.
So, what do you expect?
When parvovirus hit our dog population I had calls coming from the US for supplies of cat parvo vaccine with a two shot protocol to stop the deaths.
Never trialed.
When calicivirus escaped the containment line and wiped out millions of rabbits in Australia the TGA gave provisional approval to an imported vaccine.
It’s still provisional, it’s never been trialed.
At least vaccination is the Devil you know by now.
The Virus, it keeps morphing.
Perhaps all will be well.
In the old days the question was
‘We can put a man on the moon but cannot cure the common cold’
Perhaps, like rabies, smallpox, plague and polio we will beat this one.
However despite the TGA statement, my experience of front line medicos is that they get special clearance to use novel treatments.
My daughter was the first in Australia to have i/v infliximab infusions to treat crohn’s fistulas.
This class of drug is now widely used.
The fact that in NSW Covid cases are being extensively hospitalised probably means the medicos are still doing their job, just as they do with my daughter.
This article and the following comments are exactly why I subscribe to Quadrant. All the best to your daughter Mr Buckingham.
I attached an Oximeter and registered a healthy SpO2 of 98, put a mask on and watched it drop to 92, indicating hypoxemia, a life-threatening consequence of oxygen deprivation which can only sensibly be attributed to wearing the mask.
.
On a prior occasion I was forced to wear a mask for about 30 minutes in a Police Station. My wife says that when I came out I was terribly pale, and I walked straight into a table and chairs – maybe it’s just a coincidental, quick onset of dementia, but my mental prowess has noticeably slipped in the 10 months since.
Full marks for having the integrity and courage to speak truths about this topic. One can access such information from many alternative media sites but it is heartening to see experts in the field speaking out.
Masks are unhealthy and very poorly studied although some studies have concluded if mooted today for surgical staff they would be rejected. They remain out of tradition.
this study has some interesting material.
https://www.mdpi.com/1660-4601/18/8/4344/htm