Hydrogen power is now all the rage it seems. And with Andrew Forrest pushing it, it may eventually find a place among all the other peripheral energy sources with which we’re currently encumbering ourselves. I have not yet had a chance to delve into the pros and cons of hydrogen as a source of mains power, but I understand that its extraction is not without its problems. There are two sources, green and blue. From the website of Petrofac, which describes itself as ‘a leading energy services company that helps our clients meet the world’s evolving energy needs’:
Blue hydrogen is when natural gas is split into hydrogen and CO2 either by Steam Methane Reforming (SMR) or Auto Thermal Reforming (ATR), but the CO2 is captured and then stored. As the greenhouse gasses are captured, this mitigates the environmental impacts on the planet.
The ‘capturing’ is done through a process called Carbon Capture Usage and Storage (CCUS)…
…Green hydrogen is hydrogen produced by splitting water by electrolysis. This produces only hydrogen and oxygen. We can use the hydrogen and vent the oxygen to the atmosphere with no negative impact.
To achieve the electrolysis we need electricity, we need power. This process to make green hydrogen is powered by renewable energy sources, such as wind or solar. That makes green hydrogen the cleanest option – hydrogen from renewable energy sources without CO2 as a by-product.
But if we look at extracting hydrogen from water there is a paradox. To get hydrogen from water we separate it into its two fundamental components: two atoms of hydrogen and one atom of oxygen. That takes a certain amount of energy. When we burn hydrogen to obtain energy, we get water as a by-product – two atoms of hydrogen combined with one of oxygen. Does the amount of energy generated by the burning of hydrogen exceed the amount used to produce the hydrogen? The law of conservation of energy would suggest not.
This sounds to me rather like Snowy Hydro II, in which the system itself uses more energy than it produces and the only way it pays is that the input side of the equation is subsidised by purportedly cheap renewables. The theory being that we make the hydrogen when the sun is shining and the wind is blowing and store it to provide baseload power. I’m sure Alan Moran can address the technical and economic issues of this proposal better than I. But we won’t be storing all the hydrogen to generate electricity. A large proportion of it will be consumed all day, every day in propelling the electric vehicles so beloved of Bill Shorten.
I recently heard a story on Their ABC extolling hydrogen power EVs. Apparently, one set a new world record of some 1300 kilometres on a single tank. Pretty impressive. Part of the performance is due to the fact that the hydrogen tank and its contents is much lighter than the conventional batteries currently being used in electric vehicles. The hydrogen in the ABC’s cited example is burnt to charge a single battery. And, of course, the longer you drive the lighter you become. Sounds pretty good. But there is one glitch that occurs to me. What about the exhaust? According to Cars Guide:
Hydrogen stored in the hydrogen fuel tank reacts with oxygen in the fuel-cell stack and produces electrical energy, heat and water. The heat and water goes out the exhaust pipe as water vapour – completely harmless, as opposed to the carbon dioxide that’s released by cars with internal-combustion engines – and the electricity either goes straight to the electric-hydrogen motor or to a battery that stores the electricity for when the vehicle requires it.
But wait, I hear you cry. Water vapour completely harmless? Aren’t we told by the IPCC that water vapour is a more powerful greenhouse gas than CO2? Indeed, is not the theory that warming will become catastrophic, not because of the CO2 itself but because of the amplification to that warming produced by increased water vapour in the atmosphere? Here’s what NASA says:
Water vapor is known to be Earth’s most abundant greenhouse gas, but the extent of its contribution to global warming has been debated. Using recent NASA satellite data, researchers have estimated more precisely than ever the heat-trapping effect of water in the air, validating the role of the gas as a critical component of climate change.
Andrew Dessler and colleagues from Texas A&M University in College Station confirmed that the heat-amplifying effect of water vapor is potent enough to double the climate warming caused by increased levels of carbon dioxide in the atmosphere.
Do we know how much water vapour will be released into the atmosphere as a result of millions of hydrogen powered EVs going on 1300km trips? And how much of that solar and wind power, which would otherwise be producing electricity just to keep our lights on, will be diverted to making hydrogen? Just asking.
 Sign In
Sign In 0 Items (
0 Items ( Search
Search










Oh the hilarity of it. First we release oxygen to the atmosphere when the common view is that a tiny amount of human caused CO2 converts if from a harmless gas to a planet destroying danger. Then we contemplate the same with oxygen. In a centuries’ time will Greta’s grandkids be wringing their hands in furious alarm over the danger of anthropogenic oxygen making oxidation more potent? And then the water vapour that Peter has nicely dealt with! We’ll be spiraling out of sanity once more.
‘the heat-amplifying effect of water vapor is potent enough to double the climate warming caused by increased levels of carbon dioxide in the atmosphere…’ Relax, Peter. The climate warming caused by increased levels of carbon dioxide in the atmosphere is NIL. Total CO2 in the atmosphere ran its race as a global-warming gas long ago at lower levels than now prevail.
If we are to have a rational debate on gradual energy source transition (essential whether for AGW reasons or not), we need to exclude the irrational Greens and other scientifically and technologically illiterate activists/rent-seekers, whose detailed plans consist of concatenated slogans and wishful thinking about future technologies. It’s only 28 years to 2050, and we may not have our first submarine by then!
“Green” (sic) hydrogen via electrolysis is a chimera, wasting 70-75% of the electricity used to create it, and is only a champion with rent-seeking taxpayer subsidy junkies. At the moment, when every electron becomes increasingly more precious, we should use the electricity directly. More sensible solar splitting with catalysts is gaining ground, but is decades away from large scale commercialisation, and would require large areas dedicated to its generation.
Wind and solar have been proven in the real world (Europe, UK, Australia, US) to be hopeless in a dispatchable grid, which leaves nuclear (congratulations France, Korea, Switzerland) and hydro (Norway, Portugal) as the realistic frontrunners, with hot rock thermal (depending on location) and hydrogen as dark horses. We don’t have the height or water necessary for continent-wide hydro. Deep earth thermal is great for those countries on a tectonic plate boundary, so that leaves us out. Nuclear is the only option for us if we are dumb enough to eschew coal and gas (and it seems we are, alas).
The technology for grid connection for wind and solar is not here yet, and let me remind you again it’s only 28 years to 2050. Unfortunately, we are also dumb enough (those subsidies sure must have been juicy and irresistible) to choose to first implement wind and solar into a dispatchable grid, and disengaging from this idiocy will be ugly, and costly.
Further reading. 1. Why a hydrogen economy doesn’t make sense (www.phys.org, 2006)
2. Hydrogen the once and future fuel (John Constable, GWPF Report 44, 2020.)
3. Making clean hydrogen is hard, but researchers just solved a major hurdle (www.phys.org, 2021)
I read a couple of long articles on this during the week and the conclusion I drew from both was that its advocates don’t care about money.
“lts advocates don’t care about money.” Only the Greenies. The smart boosters care only about money ie the vast cornucopia of public subsidies that have made people like Elon Musk very rich indeed.
Twiggy has crossed the Continent to chum up with Anastasia with the idea of setting up a hydrogen plant in Gladstone.
It’s a good choice; makes a lot of sense — Queensland still has some good coal-fired power stations to provide all the electricity needed to split the water into its components by either electrolysis or SMR.
There is only one way to think of ‘blue’ and ‘green’ hydrogen; Dumb, and Dumber.
Sorry to upset you lot but you really need to see that coal exports are going to stop. Thermal coal anyway. I would also stop coal used to reduce iron ore and use it all here but as you all found hydrogen can do that job so its exports may stop as well. Hence we will have no money from coal exports. And they give us a great deal of money. The world will be going to H2. They will not like blue H2. They will want green H2. Hurry up and realise Twiggy is doing his bit to keep us all in money, not just him.
And of course the energy expended in compressing the hydrogen to a useful volume will further degrade the energy efficiency of green hydrogen, by a great deal I imagine. Because it will have to be compressed into holding chambers at point of production, then pumped into trucks, then pumped into charging stations, then pumped into vehicles. And as well as a nationwide network of charging stations we’ll also need a network of hydrogen pumping stations.
Peter O’B, doesn’t ammonia have to be created to enable the hydrogen to be transported?
Brian Boru,
possibly. I need to investigate this further
This para might help:
“Converting hydrogen into ammonia only to convert it back again might seem strange. But hydrogen is hard to ship: It has to be liquefied by chilling it to temperatures below −253°C, using up a third of its energy content. Ammonia, by contrast, liquefies at −10°C under a bit of pressure. The energy penalty of converting the hydrogen to ammonia and back is roughly the same as chilling hydrogen, Dolan says—and because far more infrastructure already exists for handling and transporting ammonia, he says, ammonia is the safer bet.”
@maxpart27
>”Sorry to upset you lot but you really need to see that coal exports are going to stop”
You have no idea. You really don’t.
For credibility, you need to expound on how China, South-East Asia, India, Taiwan, South Korea, Japan, Vietnam, Cambodia … are going to stop buying Newcastle thermal and trash their power generation within a decade or four. That’s a real question, not an arm-waver.
Then you need to expound exactly on the practical aspects and economics of using electrolysis on a gigantic scale to supply, transport and then reduce iron oxides to iron within a decade or five.
Magic, perhaps. We see lots of silly rhetoric, such as your comment, but never any hard, sensible expositions. Just technobabble.
Any country with hills and water, or sunshine and water, or nuclear power and water, can produce hydrogen by electrolysis. The idea that we will be able to make it, store it and ship it to another country (eg Japan) cheaper than they can make it themselves is a scam and a fantasy.
Adeleagdo
Your comment reminds me of a short conversation I had back in late 2009.
It was with a Tasmanian Treasury official at the opeining of the TVPS facility recently acquired by the Stategovenrment. He said that the new power station would generate lots of revenue for th State by importing gas from Victoria and sening the ‘value added’ electiricty back to Victoria.
I replied that if that were to work, they might try importing wheat from Western Canada, milling it, and sending the flour back to Ontario.That was followed by………….silence.
Count Ferdinand von Zeppelin was one the first to use hydrogen on a large scale. It proved rather dangerous.
Some of the energy available from coal combustion comes from the hydrogen component of coal. Shall we call it “black” hydrogen?
There is roughly one atom of hydrogen for each atom of carbon in a thermal coal. Geoff S
Excellent piece on one element of this nonsense.
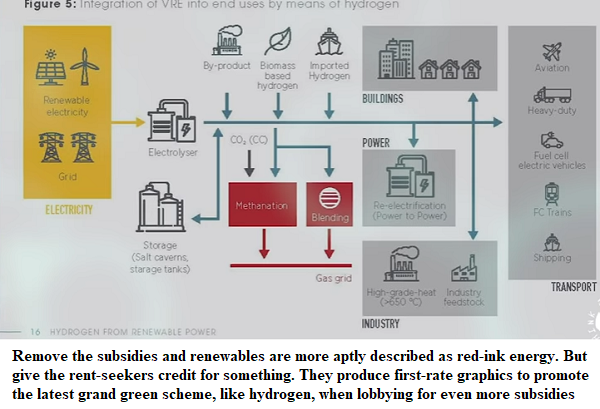
The capacity of green carpetbaggers to produce misleading diagrams, names and slogans is both wonderful and hilarious.
On the graphic heading the article, showing the initial panel of solar and wind powering the electricity grid, they should add a big red cross through the two towers signifying the grid, as we already know that ‘renewables’ destroy synchronous despatchable power on the grid; they don’t use it.
Then there is the hilarious reference to carbon capture and storage preventing intermediate CO2 emissions – the same CCS that has been a failure in more than a decade of effort to run at commercial scale. This is magical thinking, solving an unsolvable problem by just incanting a magic phrase.
And as for ‘blue hydrogen’ and ‘green hydrogen’, as distinct from ‘Hindenberg hydrogen’, it’s just a joke.
But this farrago of nonsense is wind in the sails of the green left, and PM Scott Morrison. It really appeals to his ‘Scotty from Marketing’ past.
But how efficient is it to produce Hydrogen from water with electrolysis? Just running a current through water, such as in an overcharging flooded-cell lead-acid battery, doesn’t have much of a rate of gas generation. There may be special technologies using platinum-metal catylists, but why don’t I see small scale units for sale on the market? I would buy one and use the surplus electricty to generate hydrogen for use in cooking, to replace having to fill up gas bottles with LPG. But, how much energy is consumed to cook my meal, which includes brown rice? The feasability of these things have to be worked out on a micro-scale.
ianl
You nailed it in your last para “Magic, …….. [and w]e see lots of silly rhetoric, …………… but never any hard, sensible expositions. ……… [on the facts of the matter].”
The simple fact is this:
The total energy equation contains so much ADDITIONAL energy on both sides (what goes in must equal what goes out – law of conservation of energy) that any surplus energy for useful endeavours, like cars, trains, and automobiles, is dwarfed by the wasted energy in the production, storage, distribution, and the practical use of ‘blue’, ‘green’, or ‘black’ hydrogen that even brainless morons would see it’s only ever about the money, nothing else.
And, we the people have to pay for all the gullible politicians making these stupid decisions as they want a slice of the action too, never minding that the plebs pay for it all in higher costs of living that make no common sense at all.